There is a difference between stereoselectivity and stereospecificity. Stereoselectivity means the selectivity of the product while stereospecificity means the selectivity of the reactant. if any molecule on reaction gives two stereoisomers like cis-trans or D and L or R or S then the reaction is said to be a stereoselective reaction. It is possible that one of the isomers is in large quantity (major product) as compared to other isomers. These are further divided into two groups:
Types of Stereoselectivity
- Diastereoselective reaction
- Enantioselective reaction
Diastereoselective Reaction
If two products formed are diastereomers (cis and trans), then the reaction is a diastereoselective reaction.
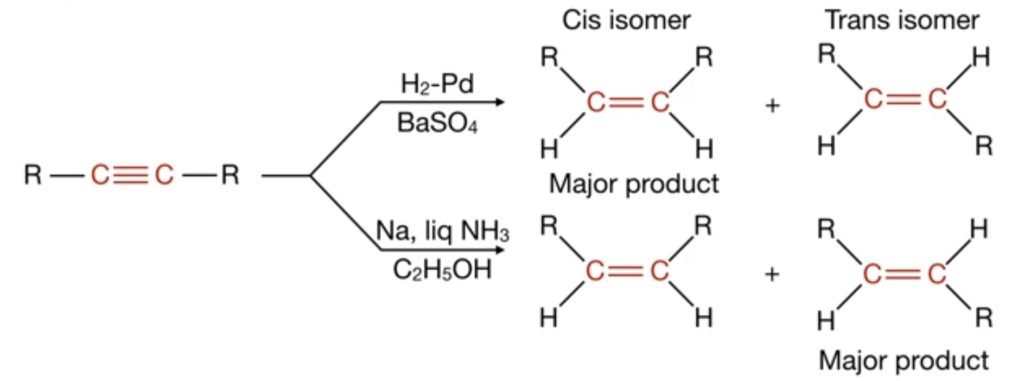
Hydrogenation of alkyne in the presence of H2-Pd/BaSO4 produces both the isomers ( cis-trans) but cis-isomer is the major product. Similarly, if the reaction is carried out in the presence of Na, Liq NH3/C2H5OH, both the isomers (cis-trans) are produced but this time trans-isomer is the major product. That’s why it is a diastereoselective reaction.
Enantioselective Reaction
If the product is enantiomers (non-superimposable mirror image) then it is an enantioselective reaction.
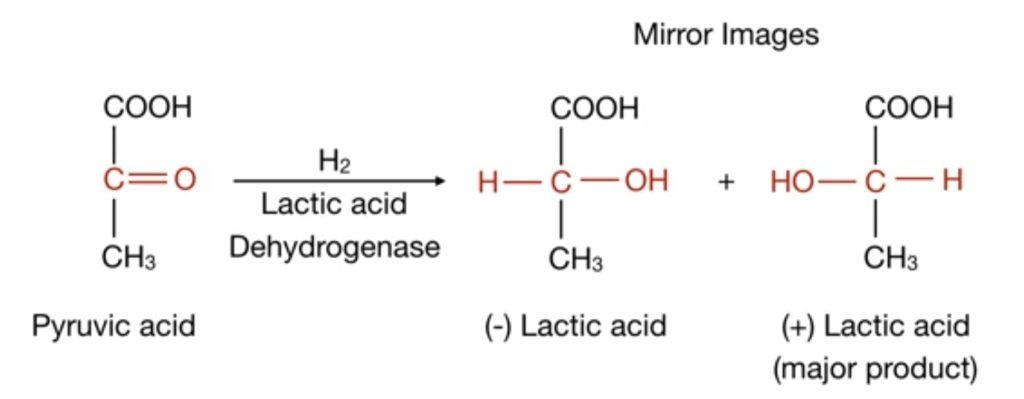
Hydrogenation of pyruvic acid produces (-) lactic acid and (+) lactic acid. These are the mirror image of each other and are called enantiomers but (+) lactic acid is a major product. so this reaction is an enantioselective reaction because it produces two enantiomers.
Chemoselective Reaction
A reaction that operates on one functional group in the presence of other functional groups is said to be a chemoselective reaction.


In the above example, when the compound is reduced with Lithium Aluminium Hydride (LiAlH4) and the hydronium ion, it reduces both the carbonyl groups so, this is not a selective reaction for one functional group. But if the reaction is carried out in the presence of sodium borohydride (NaBH4) and ethyl alcohol (CH3CH2OH), it reduces only one carbonyl group and the other carbonyl group remains unaffected. Thus, this reaction is specific to only one functional group out of two so it is a chemoselective reaction.
Regioselective Reaction
The meaning of regio is direction or region. If a reaction is selective to only one direction or region of the reacting molecule then we can say the reaction is regioselective.

In addition to HCl to propene, there are two possibilities, The first possibility is that chlorine can go the first carbon atom, and the second possibility is that chlorine can go the second carbon of double bond. According to Markovnikov’s rule, 2-chloropropane is formed as a major product. Thus reaction is selective to only one carbon atom and is called regioselective reaction.
Stereospecific Reaction
If different isomers of same compound give different product then the reaction is stereospecific. That means the final product is decided by the stereochemistry of the reactant.
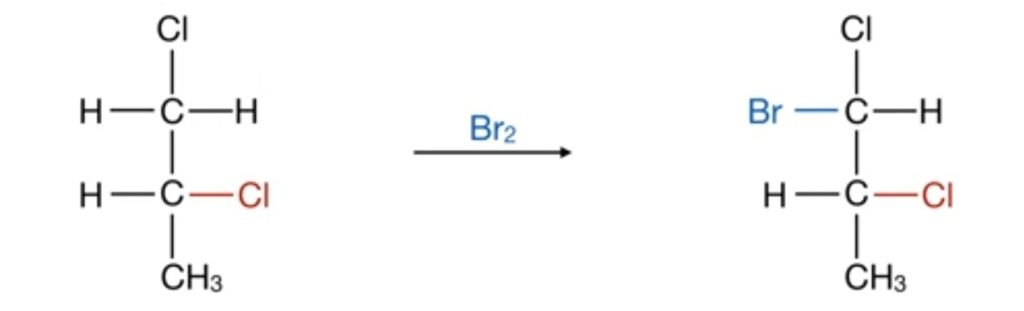
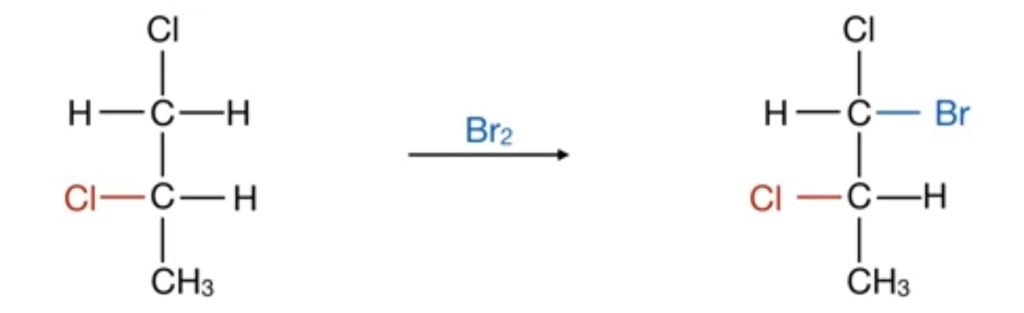
In above reactions if chloride is present on right hand side of the molecule, on bromination, bromine is attached to the left hand side of the molecule. Similarly, if the chloride group is attached on the left hand side of the molecule, the bromide group attached on the right hand side of the molecule. So the point of attachment of bromine is always dependent on the presence of chloride group. Therefore, it is stereospecific reaction.
Stereoselectivity and Stereospecificity
Let us take an example of addition reaction of alkenes, if we add bromine in 2-butene, we get 2,3-dibromobutane.

As we know that 2,3-dibromobutane has two chiral carbons. The product (2,3-dibromobutane) can exist as meso compound due to internal compensation and superimposable mirror image of each other.

But the reactant exist as diastereomeric, a pair of geometric isomers. (cis-2-butene and trans-2-butene)
After that we conclude that our reactant have different stereochemistry and our product is have different stereochemistry. So we can say that different stereochemistry of reactant gives different stereochemistry of product and this is called stereospecificity. Now the question is:
- What product will we get from cis 2-butene?
- What product will we get from Trans 2-butene?
Trans 2-butene yeild the racemic mixture as cis form? No, trans 2-butene yields only meso 2,3-dibromobutane. The reaction in which stereochemically different reactant gives stereochemically different products is called a stereospecific reaction and the process is called stereospecificity.
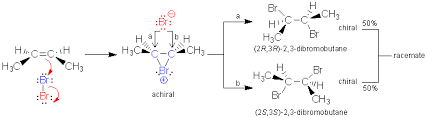
Anti-Addition of Bromine to Cis 2-butene
Cis 2-butene yields only racemic 2,3-dibromobutane, none of the meso compound is obtained because of anti addition of bromine in alkenes. A reaction that yields predominantly one stereoisomer (or one pair of enantiomers) of several diastereomeric possibilities is called a stereoselective reaction and the process is known as stereoselectivity.
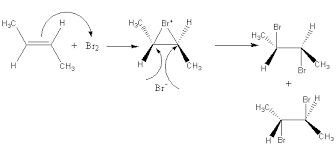
Anti-Addition of Bromine to Trans 2-butene
Trans 2-butene yeild the racemic mixture as cis form? No, trans 2-butene yields only meso 2,3-dibromobutane. The reaction in which stereochemically different reactant gives stereochemically different products is called a stereospecific reaction and the process is called stereospecificity.
- Stereoselectivity is mainly about the product.
- Stereospecificity is mainly about the reactant.
- Sometimes, stereoselectivity work for both reactant and product.
Stereoselectivity
Stereoselectivity is a product selectivity. A stereoselective reaction is one in which a single reactant can give two or more stereoisomeric product and one or more of these product is preferred over the others-even if the preference is very small.
Stereospecificity
Stereospecificity is a reactant selectivity. A stereospecific reaction in which one sterioisomer of the reactant gives one stereoisomers of the product, while a different stereoisomer of the reactant gives a different stereoisomers of the product.



Leave a Reply