Is Acetic Acid Strong or Weak?
This blog is helpful for you to understand the question Is acetic acid strong or weak? Acetic acid is a weak acid that does not completely ionize into its ions in a solution.

It is a reversible reaction which means the reaction can proceed in both directions.
An acid is any species that can donate hydrogen ions in an aqueous medium. Acids have a sour taste. Acids have PH less than 7. Acids are classified into two types on the basis of ionization ability. The acids which ionize completely in an aqueous solution is called strong acid. The acids which do not ionize completely in an aqueous solution are called weak acids.
Acetic acid is incompletely ionized in water and has a very low ionization extent about less than 10%. It has a poor ion donation ability. It is commonly called ethanoic acid, or vinegar acid. It is most commonly found in vinegar. Acetic acid is not produced more ions in water so that’s why it is a very common weak acid.
Applications:
- Acetic acid is used for the preservation of food items.
- Acetic acid is used in the preparation of acetic anhydride, plastics, vinyl acetate monomer cellulose, dyes, photographic film chemicals, and chloroacetic acid.
- Also used in the preparation of vitamins, antibiotics, organic chemicals, and hormones.
Harmful effects:
Acetic acid is an eye, and skin irritant. Prolonged used cause skin irritation due to damage to tissues. It is observed that acetic acid causes cancer in humans.
Why Formic Acid Is Stronger Acid than Acetic Acid?
After knowing the concept of whether acetic acid strong or weak, now we will find which one has a more acidic character acetic acid or formic acid? Look at the chemical structure of both:
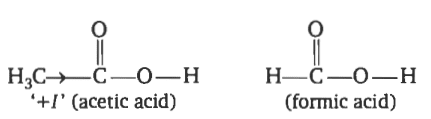
In acetic acid, the methyl group is attached and methyl is the electron-donating group due to the +I effect (Inductive effect). The methyl group is electron-donating as compared to the hydrogen atom. Thus, the acetic acid increases the electron density in the -OH group, due to this reason the -OH bond is not easily ionized. So acetic acid is 10 times weaker acid than formic acid.
Determination of Acetic Acid In Vinegar
How to determine the percentage of acetic acid in vinegar by using lab apparatus.
Apparatus or Reagents Required
- NaOH solution
- White vinegar
- Potassium hydrogen phthalate (KHP)
- Phenolphthalein indicator
- Buret
- Wash bottle
- Stand
- Erlenmeyer flask
- Pipet filler
- Pipet (10ml)
- Funnel
Part A: Standardization of NaOH Solution
- Rinse the buret with distilled water
- Set up the titration apparatus
- Rinse the buret with a little amount of NaOH solution and then fill it with NaOH solution.
- Record the initial reading, and make sure that there are no bubbles in the buret.
- Weight a sample of potassium hydrogen phthalate (KHP) of about 0.2g
- Place the sample of potassium hydrogen phthalate into a 250ml flask, add 50ml of distilled water and swirl until the acid is dissolved.
- Add three drops of phenolphthalein indicator to the KHP sample.
- Place the flask on a piece of white paper under the buret and allow the sodium hydroxide solution to run in slowly with swirling.
- At the endpoint, one drop of base produces a permanent pale pink color. Record the final volume of the base used.
Part B: Mass Percent of Acetic Acid in Vinegar
- Rinse the pipet with small portions of the vinegar solution and discard the solution used for washing.
- Transfer 10ml of the vinegar solution into a clean 250ml Erlenmeyer flask.
- Add about three drops of phenolphthalein indicator to the vinegar solution.
- Place the flask under the buret and record the initial reading on the NaOH buret, start adding the base to the acid solution while stirring the acid solution constantly with a rotary motion of the flask.
- At the endpoint, one drop of base produces a permanent pale pink color. Record the final volume of the base used.

Find the PH of weak Acetic acid having 0.002N. If it is 2% ionized?
CH3COOH ⇌ CHCOO– + H+
Molarity = 0.002M = C
N = M × x
0.002 = M × 1
M = 0.002
CH3COOH ⇌ CHCOO– + H+
Initially “c” “0” “0”
At Eq “c-cα” “cα” “cα”
The value of α is given in question
α = 2%
α = 2/100
α = 0.002
[H+] = cα
[H+] = 0.002 × 0.02
[H+] = 2×10-3 × 2×10-2
[H+] = 4×10-5
pH -log [H+]
pH -log [4×10-5]
pH = -log 4 + log 10-5
pH = -(2log 2 – 5)
pH = – (2×0.3 – 5)
pH = 5-0.6
pH = 4.4
Related Post
Is NaOH Acid or Base? | Manufacturing of NaOH



Leave a Reply