This blog will basically give a complete piece of information about boron trifluoride BF3. This will also explain the BF3 Lewis structure, and its structural properties as well. Structural properties explanation is based on the lewis structure. In simple words, we can say that drawing lewis’s structure is necessary to explain all the structural properties including bond angle, hybridization, geometry, etc. We should also have some understanding that what is boron trifluoride? What are its physical properties?
What do you know about BF3??
BF3 is a structural formula of boron trifluoride. Boron trifluoride shows very interesting behavior in the different states of water. It may occur in a dual state. In the liquid state, it is highly soluble because these are coordinate covalent compounds that have the ability to dissociate in water and dissolve more easily. On the other hand, if it exists in the gaseous state, it is highly toxic because of the creation of white fumes in the air.
Boron trifluoride is a colorless gas and has a strong odor. When it is converted in the vapor phase they have a high electron density. Mostly it is highly dissolved in the water and also dissolved in that solvents have oxygen and nitrogen in them. Its exposure to the skin causes the burning sensation and its long-term intact with fire causes the bursting of fire and rocketing of a container.
Borotriflouirde is considered to be an active catalyst. In isomerization, hydration, dehydration, and sulfonation it acts as a catalyst.
What are the properties of BF3??
In the above explanation about what is BF3? We will discuss its properties in detail but now we will discuss some of the additional physical properties.
- In anhydrous form its molar mass is 67.82 g/mol but if it is present in dihydrate form its molar mass is 103.8 g/mol.
- Just like molar mass, the second property may refer to density in anhydrous and dihydrate form. In anhydrous form, its density is 0.00276 g/cm3 but in dihydrate form, its density is 1.64 g/cm3.
- Its melting point is -126.8 ℃ and its boiling point is -100.3℃.
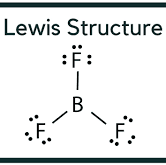
How to draw BF3 lewis structure??
Before drawing the lewis structure of BF3, we have to discuss why we draw the lewis structure?? The answer to this question is that lewis’s structure has very important to deal with the structural properties of a compound. By drawing lewis’s structure we can predict completely the arrangements and number of electrons present in an atom. By knowing the number and arrangements of electrons in an atom, it is easy for us to know the structural properties of the compound.
Rules to draw BF3 Lewis structure:
Before drawing the Lewis structure, it is necessary to understand the rule to draw Lewis structure.
- Firstly, we have to calculate the number of valance electrons in an atom.
- Then we find the number of electrons by which an atom forms a bond.
- In the third step, determine the number of bonds in both atoms.
- Now, find the central atom, i.e. Boron
- In the last place the electrons according to their positions.

As boron is less electronegative so that’s why it has a 100% chance to be an electropositive atom. It is a necessary condition for a lewis structure that the central atom must be electropositive. Since, after selecting the central atom we will draw no. of lone pairs and at last, all the electrons are placed around the central metal atom.
The polarity of BF3:
Polarity may refer to the polar or non-polar compound. Polar compounds are the compounds at which some partial charge is present but in non-polar compounds, there’s no development of poles and no development of partial charges. But, there is a condition for polar compounds that both atoms are heteroatoms.
Just like in BF3 molecule it has two atoms in which one is electropositive and fluorine is electronegative. Electropositive has the ability to donate an electron but electronegative has the ability to accept an electron. So both of the atoms share electrons and are stable. So, no one can accept and donate an electron to become unstable. So, to avoid unstability partial is developed but electrons neither accept nor donate.
So, for this purpose, it is concluded that BF3 (BORON TRIFLUORDIE) is a polar compound. This reason causes it to soluble in water due to the same polar nature of water and BF3 according to the principle of “Like dissolve Like“.
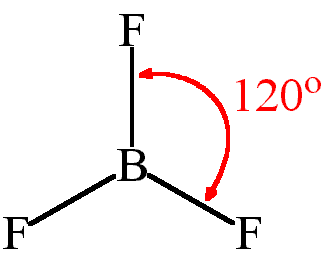
Knowing Hyberdization, bond angle & geometry of BF3:
As we know that there is only s orbital available for boron. And two p orbitals overlap with the s orbital of Boron. So, form sp2 hybridization. It makes 3 sigma bonds with boron so possesses triangular geometry with the bond angle of 120o. A very conceptual concept of geometry formation is that. In BF2 structure, there is linear geometry but when another fluorine is attached to boron, its electronic cloud produces a repulsive effect on another fluorine atom, and hence, they move in a downward direction.