
Instead of directly moving on to the detail of the state function. Let’s first have the basic concept of State function through some general examples.
Does the Basic Concept deal with What a State functions?
To answer this question first consider a lake refilled with rainwater or artificial means with the help of pipes. We have no concerns about the means by which the lake is refilled. We are looking for a change in the water level in the lake. Change is totally dependent on the initial position and final position regardless of the path by which change is brought about. Such functions are known as state functions.
Let’s have a look at the second example of state function. Suppose that Mr. Tony has a water sample and he wants to change the temperature of the water. He can change the temperature of water by different means either by heating or by lowering the temperature and his main concern is the change in temperature. In this example, Mr. Tony is taking into account the change in temperature regardless of the path by which the change is brought.
Definition of State Function
Those functions have values totally dependent on the initial and final state of the system without any influence on the path by which change is brought about.
OR
Those physical quantities which merely state-dependent regardless of the path are known as a state functions.
Examples for State Function
Due to any reason or by any means change in the system occurs. These changes are in terms of volume, pressure, and temperature. Also known as the variables of the state. Some important variables of the state are listed down.

How volume, temperature, and pressure are state functions?
- Temperature is basically the amount of kinetic energy possessed by the system in simple words it is the hotness and coolness of the system and represented by (T). Now change in the temperature of the system is totally dependent on the initial and final state. As like ΔT = T(F) – T(i) . Change in temperature is on the behalf of final minus initial temperature so it is a state function.
- Volume is the physical occupation of space by a substance or in other words space taken by the matter. It is also a state function because any change in it totally relies on the initial and final position of the system. The same is the case no path influences init. ΔV = V(F) – V(i)
- Pressure is, in general, the force on the unit area ( P= F/A ) or thermodynamically it is the force applied by molecules in-cylinder per unit area on the walls of a container. Any change in volume is dependent on the initial and final state of the system. As ΔP = P(F) – P(i) from the equation it is clear any change in it will measure by its initial and final values.
Internal Energy is a State function how?
Internal energy in simplest form is the total energy of the system or The sum of all the type of energies present in the system.
It is a state function because it is totally dependent upon the system state regardless of how the system brought the state. Any change in the internal energy is in the final and initial position of the system. Internal energy change is chemically given as
ΔU = U(Product) – U(Reactants)
From the equation, it is clear that in a reaction any change in internal energy is state-dependent. If the U1 is greater than U2 in this case system will release energy and then ΔU = -ve. If the U1 is greater than U2 in this case system will absorb energy and then ΔU = +ve.

How does Enthalpy a state function?
Enthalpy of a system is the sum total of internal energy and PV ( pressure and volume) , in the some conditions.
Enthalpy is a heat content and H is for the representation of enthalpy.
H = U + PV
Let’s consider an enthalpy of a chemical reaction whose change depends on the initial and final values. Thus the change in enthalpy is state-dependent not a path, so it is a State function.
ΔH = H(Product) – H(Reactant)
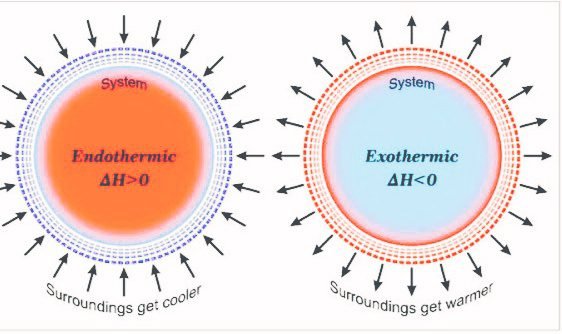
When reaction releases the energy then the Enthalpy of the system is negative and when reaction absorbs energy then the Enthalpy of the system is positive.
Entropy is a state function, is it?
It is define as the disorder and irregularity of the system. OR In other words it is measuring of disorder ness of the system.
Gas generally has more entropy than liquid and solids.
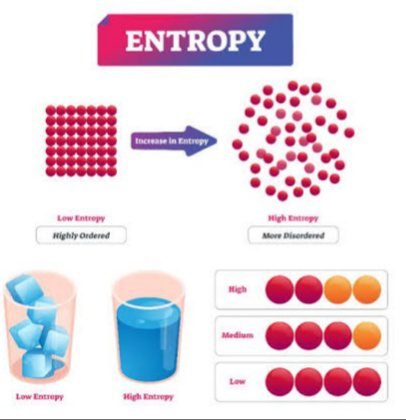
Factors that affects the Entropy
- Temperature
- volume
- Physical State
These are most ruling factors on entropy and all these are state-dependent. Thus entropy is also a state function because it depends on the initial and final state of system disorder ness.
What’s the key difference between state and path functions?
| State Function | Path Function |
| It totally depends on the initial and final state | It has no concerns with the initial and final state of the system. |
| State functions have no concerns with the path adopted by the system in bringing the change. | Path functions are totally dependent on the path adopted by the system. |
| State function has cyclic integrals zero. | Path functions do not have their cyclic integrals zero. |
| Example: Enthalpy, Entropy, Volume | Example: Heat, work |



Leave a Reply